Author: Matthew Perrone
-

First over-the-counter birth control pill in US begins shipping to stores
The first over-the-counter birth control pill will be available in U.S. stores later this month
-
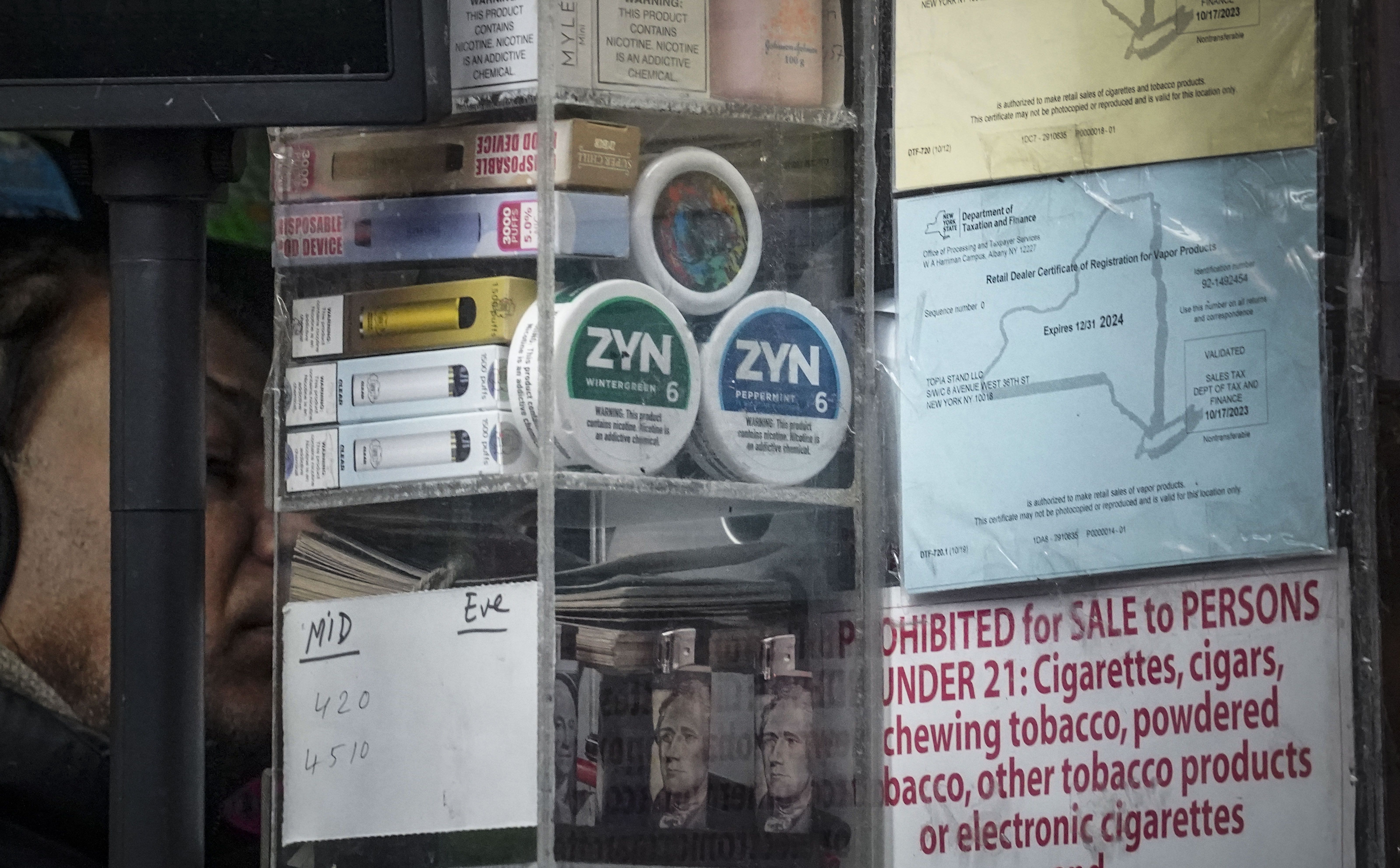
Zyn nicotine pouches are all over TikTok, sparking debate among politicians and health experts
A small nicotine pouch called Zyn has sparked a big debate among politicians, pundits and experts
-

As investors pile into psychedelics, idealism gives way to pharma economics
—
by
Money is pouring into the fledgling psychedelic medicine industry, with dozens of startup companies vying to be among the first to sell mind-expanding drugs for depression, addiction and other mental health conditions. While psychedelics are still illegal under federal law, companies are jostling to try and patent key ingredients found in magic mushrooms, ayahuasca and…
-
Maker of recalled sleep apnea machines agrees to halt sales in U.S.
—
by
WASHINGTON • The company behind a global recall of sleep apnea machines said Monday it will stop selling the devices in the U.S., under a tentative agreement with regulators that could cost the manufacturer nearly $400 million. Device maker Philips has recalled more than 5 million pressurized breathing machines due to risks that their internal…
-
New blood donation rules allow more gay men to give in U.S.
—
by
WASHINGTON • Gay and bisexual men in monogamous relationships can give blood in the U.S. without abstaining from sex under updated federal health guidelines that focus on donors’ behavior, not their sexual orientation. The Food and Drug Administration guidelines finalized Thursday ease decades-old restrictions designed to protect the blood supply from HIV. The agency announced…
-
ALS drug wins FDA approval despite questionable data
—
by
WASHINGTON • A much-debated drug for Lou Gehrig’s disease won U.S. approval Thursday, a long-sought victory for patients that is likely to renew questions about the scientific rigor behind government reviews of experimental medicines. The Food and Drug Administration approved the drug from Amylyx Pharmaceuticals based on results from one small, mid-stage study in which…
-
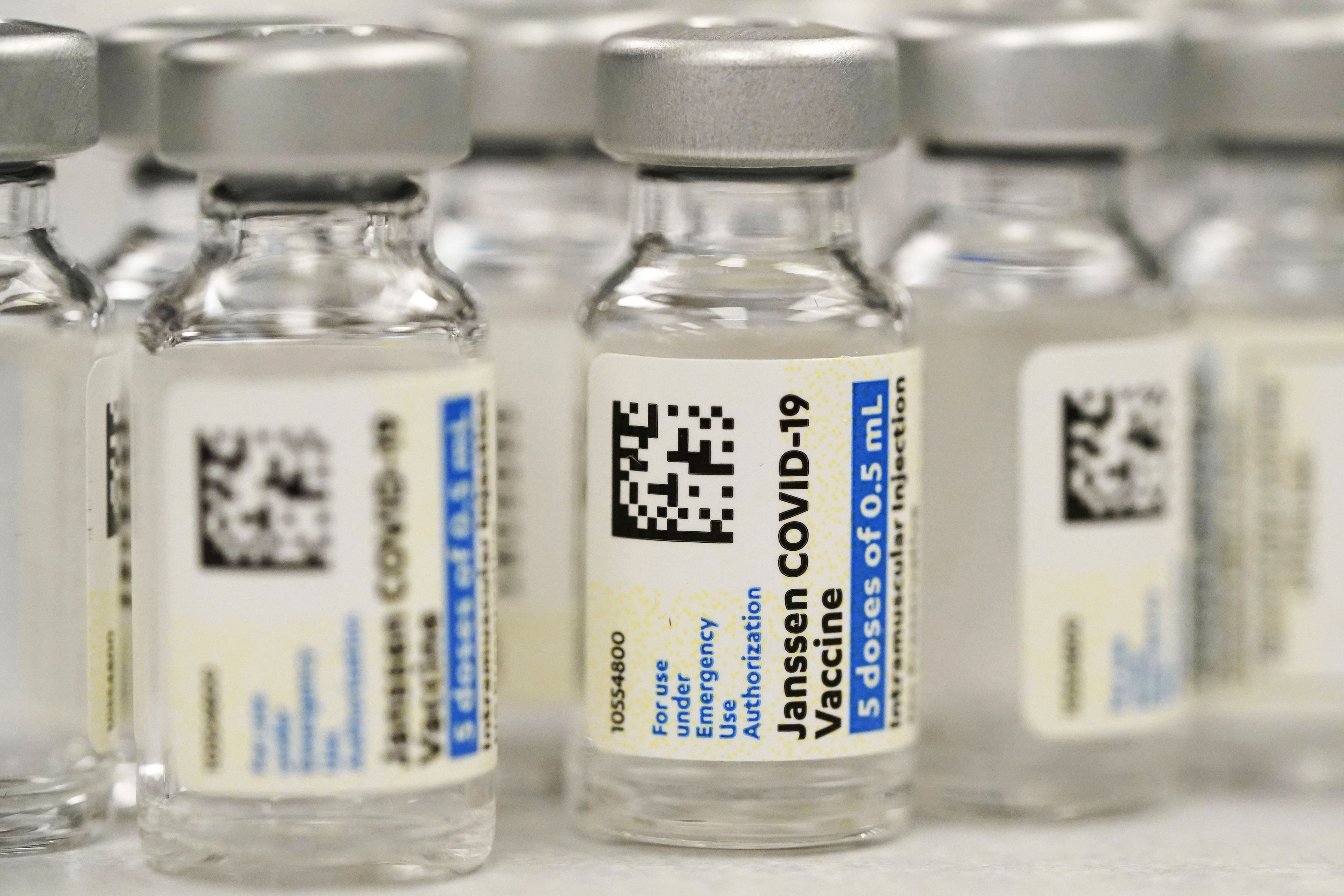
FDA restricts J&J’s COVID-19 vaccine due to blood clot risk
—
by
WASHINGTON • U.S. regulators on Thursday strictly limited who can receive Johnson & Johnson’s COVID-19 vaccine due to the ongoing risk of rare but serious blood clots. The Food and Drug Administration said the shot should only be given to adults who cannot receive a different vaccine or specifically request J&J’s vaccine. U.S. authorities for…
-

Lawmakers scrutinize McKinsey’s opioid, FDA consulting work
—
by
WASHINGTON • House Democrats vowed to continue investigating consulting giant McKinsey’s work with opioid drugmakers after a Wednesday hearing detailed how the firm had advised companies pushing painkillers as well as U.S. health regulators. The hearing before a House committee is part of a probe into McKinsey’s role in the U.S. opioid crisis that has…
-
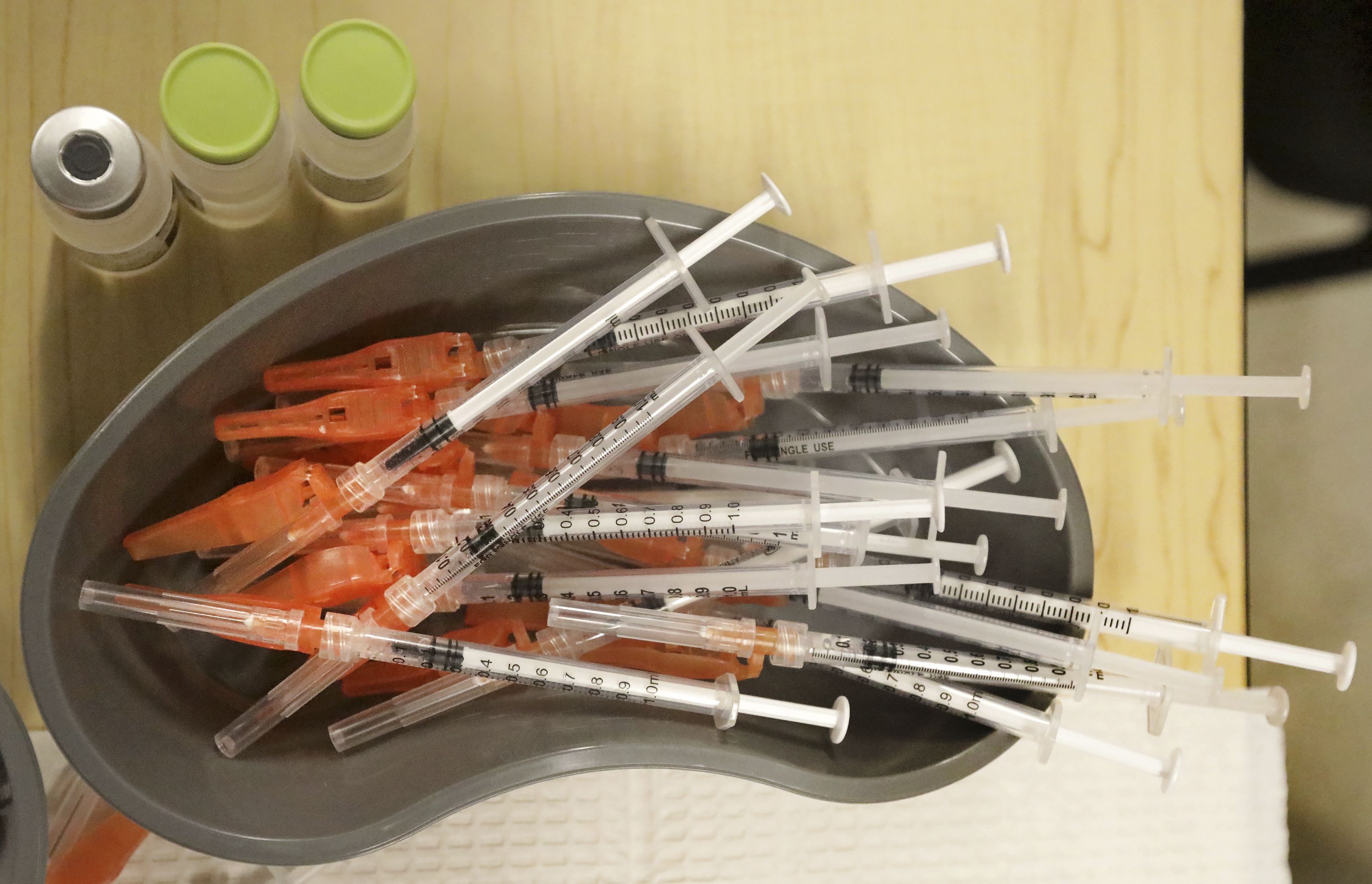
U.S. experts discuss COVID boosters for the fall and beyond
—
by
WASHINGTON • While many Americans are trying to move on with their lives after two years of the COVID-19 pandemic, U.S. health officials are debating the best way to use vaccines to stay ahead of the coronavirus. A panel of U.S. vaccine experts was meeting Wednesday to discuss key questions for future COVID-19 booster campaigns.…
-

Big drop in US teen vaping seen with COVID school closures
—
by
WASHINGTON • Teen vaping plummeted this year as many U.S. students were forced to learn from home during the pandemic, according to a government report released Thursday. U.S. health officials urged caution in interpreting the numbers, which were collected using an online questionnaire for the first time. But outside experts said the big decrease in…







